My research involves hydrogen, so by now I’ve given a spiel explaining the whole point of hydrogen a couple times. I’ve had to explain it to myself too: at the start of my research, I made a PowerPoint to organize all the basic facts for myself. This is a written-up version of that, intended as an accessible overview of the hydrogen situation for anyone unfamiliar. It goes into less disorientating detail than the Wikipedia page does, but is more comprehensive than the short EIA intro page. If I introduce some new concepts, I’ll explain them. Opposingly, I will purposefully avoid jargon where it would bloat the writing too much.
What is Hydrogen?
Hydrogen is the lightest element, defined by having only one proton. Bigger atoms are basically made by smashing together smaller atoms, so it makes sense that over 90% of all atoms in the universe are hydrogen. On Earth, 99.98% of all hydrogen atoms are the isotope 1H, which means there are no neutrons. Hydrogen atoms like to pair up into H2, which is a gas at any temperature above -240 °C or pressure below 13.3 bars (~13 times atmospheric pressure!). So, if you’re alive and hanging out with some H2, that H2 is going to be a gas.
However, there’s almost no naturally occurring H2 on Earth. Only 0.00005% of the Earth’s atmosphere is H2. You see, hydrogen is so light that Earth’s gravity isn’t always enough to hold it down. Hydrogen molecules at the top of the atmosphere can reach escape velocity and shoot off into space. So, our atmospheric hydrogen supply has diminished over Earth’s history to almost nothing.
Making H2
Thus, while we can purify the more abundant gases, like N2, O2, and even argon from plain air, we get H2 through other means. The current cheapest method, which accounts for about half of all H2 produced, uses natural gas. Natural gas is mostly methane (CH4), which reacts with steam (H2O) at a high temperature (~1000 °C) in the presence of catalyst to form carbon monoxide (CO) and H2. We can then react that CO with more H2O to make CO2 and more H2!
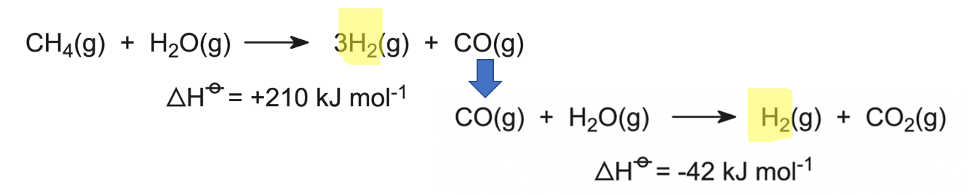
About a quarter of H2 is made from the other fossil fuels: coal and oil. The chemical processes are, with high-temperature reaction steps to break these big coal and oil molecules into H2, CO, and CO2. We always end up with that pesky CO2.
CO2 emissions are a major downside of fossil-fuel based H2 synthesis. If the CO2 just gets released into the atmosphere, then the H2 that was made is classed as grey hydrogen. If the CO2 is captured and stored away for the long-term, the H2 is called blue hydrogen. Grey hydrogen accounts for virtually all of dedicated hydrogen production, while blue hydrogen makes up only a tenth of a percent and green hydrogen, three-hundredths of a percent.
Green hydrogen is H2 made without directly producing any CO2. Instead of using fossil fuels as a source of hydrogen, it splits water, converting H2O to H2 and half as much O2. But how do we split water without consuming limited resources or producing CO2? We could try heating H2O alone until it starts splitting: thermolysis. That’s not the best option because H2O is really stable, so we’d need to heat it to far hotter temperatures than we did natural gas—like 3000°C to split about half of the water. For comparison, iron melts around 1800 °C.
Alternatively, we can include other chemicals in reactions which produce H2 from H2O at a cooler but still very hot range of 500-1800 °C[1]. A variety of such processes called thermochemical cycles have been proposed over the past century, but they’re basically all still in development. Part of the difficulty in scale-up is getting those high temperatures with renewable energy. Electric arc furnaces can technically reach the top of this range, but ones that hot aren’t commercially available yet[2]. Another option is nuclear process heat, which skips the electricity step. But don’t forget, nuclear isn’t technically renewable[3] and has its own problems.
The most widely known and used method for making green hydrogen is electrochemical water splitting, or electrolysis of water. By applying an electric potential across two electrodes immersed in water (with some other stuff to help out), we can produce H2 at one and O2 at the other.
| Energy Form | H2 Source | Other Key Materials | Method | Pros | Cons |
| Heat | Water | Catalysts | Thermolysis | Simple, no toxic chemicals | Unfeasibly high temp, must separate H2 and O2 |
| Heat | Water | Other chemicals | Thermochemical cycles | Colder than thermolysis | Technical challenges remain, still needs high temp heat source like nuclear |
| Electricity | Water | Catalysts | Electrolysis | Uses electricity | Uses a lot of electricity |
| Light | Water | Photocatalysts | Photocatalytic water splitting | Uses light, simple setup | Much R&D to be done on everything[4] |
Electrolytic processes can produce things other hydrogen and oxygen. In fact, most of the H2 made by electrolysis is not through water-splitting (<0.1%), but as a byproduct of the chlor-alkali process (2%)[5].
Chlor-alkali reaction
2NaCl + 2H2O → Cl2 + H2 + 2NaOH
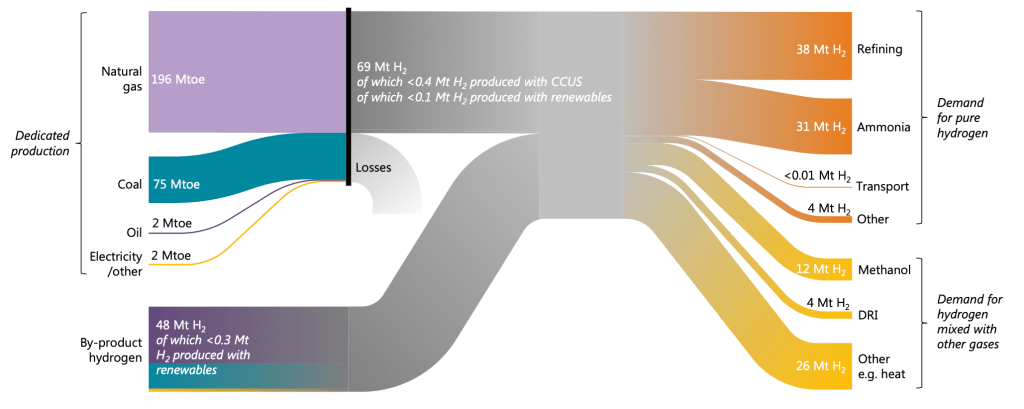
So what’s actually going on in the world? From this figure, we see about 60% comes from natural gas, another 23% from coal, and only a tenth of a percent of hydrogen is produced with renewables. Hydrogen production is not a clean business right now.
Uses of Hydrogen
The right half of Figure 1 brings us to the second topic: the use of hydrogen. We’ve got 33% for refining (removing sulfur from) crude oil, 26% for producing ammonia (mostly used in fertilizer). There’s the 23% produced as a byproduct and used for “other” purposes like heating, 10% to make methanol (mostly used to make other chemicals), and then some minor uses.
One reaction that happens in oil refining
C2H5SH + H2 → C2H6 + H2S
Producing ammonia for fertilizer with the Haber process
N2 + 3H2 → 2NH3
One reaction that produces methanol
CO + 2H2 → CH3OH
Green hydrogen can help decarbonize these industries. But none of these things are energy, and this post is about energy. The only energy purpose on this chart is transport, which uses about a hundredth of a percent of the produced hydrogen. All of the hype we hear about a hydrogen-fueled future is about precisely that: the future. But before we get into exactly how big hydrogen fuel is right now, let’s get clear about what it is.
Hydrogen as Fuel
What is the big deal about hydrogen in renewable energy? So far, we’ve only seen that it takes a lot of energy to produce. To be clear: you can’t get out more energy than you put in. Efficiency is always less than 100%. What makes hydrogen worth making is that we can get some of that energy back out in a different way, time, place, and rate.
As we already discussed, it takes energy to split H2O to H2 and O2. Doing the opposite—reassembling H2 and O2 to H2O—releases that exact same amount of energy (all else held equal). Depending on how H2 and O2 are reassembled, that energy can be released in different forms. The simplest way to do that is combustion.
H2 + O2 → H2O
When the temperature and concentration of H2 and O2 are within certain bounds, if a little bit of ignition energy is supplied via a spark, the reaction will get over its activation energy hump and occur. The reaction releases more energy as heat, which then sets off neighboring molecules, leading to a flame that burns until it runs out of fuel.
If we made that hydrogen via say, solar-powered electrolysis, we’ve converted intermittent electrical energy to storable chemical energy, which can later be converted to thermal energy.
But what if we want electricity out, not heat? We could convert that heat energy to electrical energy the way we’ve doing for ages: a gas turbine. Or, we could skip the heat step and use the mirror image of an electrolytic cell: a fuel cell. Fuel cells have H2 on one side and O2 on the other, forcing electrons from the H2 to go through a circuit to complete the formation of H2O. The theoretical maximum and practical efficiency of fuel cells is a good chunk greater than that of internal combustion engines,[6] something like 60:100 and 20:40, respectively.
Notice that if we have a flow of electrons going to one side, charge neutrality must be maintained by either a positive flow to the same side or a negative flow to the opposite side. The choice of that charge carrier and the electrolyte it moves through differentiates the main types of fuel cells.

In an alkaline fuel cell (the earlier kind, invented in 1932 by none other than Francis Bacon), an O2 joins with 2H2O and 4 electrons at the cathode to make 4OH-, which travels through some water to get to 2H2 at the anode, where they combine and release 4 electrons. That water must be thick with OH- ions (hydroxyls) to keep things moving along, so a very concentrated alkaline solution is used, hence the name “alkaline fuel cell.” These were commercially beat out by the 2000s by PEM fuel cells, but their claim to fame is powering NASA’s Apollo missions and Space Shuttle program.

For the polymer electrolyte membrane (PEM) fuel cell, H+ rather than OH– are shuttled between the electrodes to account for the charge flow. They move through a polymer electrolyte membrane that allows H+s but not electrons through, and combine with O2 and electrons to form H2O. The advantage of PEM fuel cells over other fuel cells is that they’re smaller and lighter, so more suited to transportation. PEM fuel cells also have a claim to space fame, having been used in NASA’s even earlier Project Gemini, and power most hydrogen cars on the market.

The last major kind of hydrogen fuel cell is the solid oxide fuel cell (SOFC). In this case, the electrolyte is actually a solid that conducts negatively charged oxygen ions (O–) rather than H+ or OH–. The O–s travel through the oxide to combine with H2, giving off 2 electrons to make H2O. This requires a pretty high operating temperature ranging 500-1000 °C, but the overall efficiency is pretty advantageous, with the additional perk that the “waste heat” is hot enough to be industrially useful.
There’s more types of fuel cells, such as the phosphoric acid fuel cell, but this should give you a good idea to start with.
Other Decarbonization Applications
While this post focuses on energy applications, green hydrogen also has decarbonization potential in other applications. There’s methanol and ammonia production, as I mentioned early, but also more creative applications like steel production.
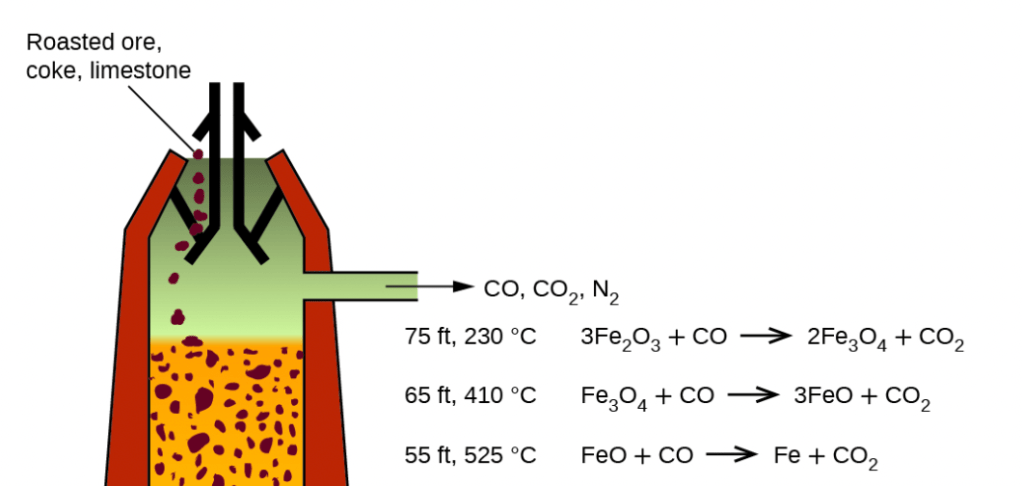
Steel is normally made by using coal as the reducing reagent to separate iron from the oxygen atoms its bonded with in naturally occurring iron ore. This reaction releases CO2. An alternative method is using hydrogen, which also loves to bond with oxygen, as the reducing agent. Read more about it green steel here.
Lastly, hydrogen is also being considered as a renewable fuel for high temperature process that might be harder to electrify, like cement kilns.
Applications of Fuel Cells
Fuel cells are useful because they provide off-grid electricity, provided you have a somewhere to keep all the H2 and O2. The O2 could come from air (no storage needed), or for touchier fuel cells, from tanks of pure O2. The main current applications of fuel cells are:
- Cars (PEMFC)
- Buses (PEMFC)
- Energy storage for businesses and homes (SOFC, PEMFC)
- Spacecraft (AE)
Less developed ideas are:
- Trains
- Aircraft
- Ships
Hydrogen is an interesting fuel to compare to oil. It’s energy-to-mass ratio is bigger, but it’s energy-to-volume ratio is smaller. It’s also a tiny molecule, which means it not only escapes our atmosphere but escapes containers. That’s why we can’t just repurpose natural gas pipelines for pure hydrogen. Thus, methods of storing hydrogen are also an active area of research and development.
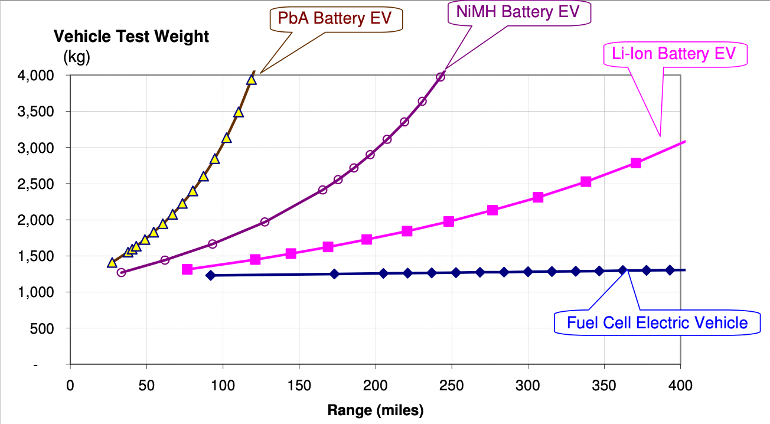
There’s also the comparison between hydrogen and battery-electric vehicles. Here, the main advantage of hydrogen is that increasing range requires only a little bit of increased weight. Fuel cells, are generally bigger than batteries, but at large capacities, are lighter than batteries. So while hydrogen systems are less efficient on a pure energy basis, at heavy and long-range enough vehicles, where lugging the energy storage around is part of the equation, hydrogen may beat out battery-electric in overall efficiency by being lighter. A disadvantage is that hydrogen transport and fueling stations are harder to build out than electric charging stations.
Current Players in the Field
Here is a non-comprehensive list of companies working on green hydrogen production and fuel cells.
- Green hydrogen
- Air Liquide
- Air Products
- Hydrogenics (owned mostly by Cummins)
- Messer
- Linde
- Fuel cells
- Ballard
- Toshiba
- Plug Power
- Cummins
- Mitsubishi
- Bloom Energy
- FC Vehicles
- Toyota
- Honda
- Hyundai
- Daimler
- Nikola
- Hyzon

To give a geographic sense of where the action is, most hydrogen cars are in California, Japan, South Korea (a more recent push), and Europe. California is the only U.S. state really pursuing hydrogen, so it’s the only state with hydrogen refueling stations and thus, hydrogen cars. Japan has a national hydrogen strategy that continues to support development. The European Commission also has published a hydrogen strategy.
Future of Hydrogen
It appears to me that there are two main promising applications for hydrogen energy systems: renewable energy storage for mid to large scale facilities and renewable-powered heavy long-range transport. These are both important problems for renewable energy that hydrogen solutions seem competitive for, as opposed to cars, where battery-electric seems advantageous on most counts. Many technical and economic problems remain, such as affordable hydrogen infrastructure, safe and scalable storage, so work on these has potential for enabling certain capabilities in the post-fossil world.
[1] Safari, F. & Dincer, I. A review and comparative evaluation of thermochemical water splitting cycles for hydrogen production. Energy Conversion and Management 205, 112182 (2020).
[2] How electrification can help industrial companies cut costs | McKinsey. https://www.mckinsey.com/industries/electric-power-and-natural-gas/our-insights/plugging-in-what-electrification-can-do-for-industry.
[3] How long will the world’s uranium supplies last? Scientific American https://www.scientificamerican.com/article/how-long-will-global-uranium-deposits-last/.
[4] Villa, K., Galán-Mascarós, J. R., López, N. & Palomares, E. Photocatalytic water splitting: advantages and challenges. Sustainable Energy Fuels 5, 4560–4569 (2021).
[5] IEA (2019), The Future of Hydrogen, IEA, Paris https://www.iea.org/reports/the-future-of-hydrogen
[6] Fact Sheet | Fuel Cells | White Papers | EESI. https://www.eesi.org/papers/view/fact-sheet-fuel-cells.
[7] Hydrogen Storage. Energy.gov https://www.energy.gov/eere/fuelcells/hydrogen-storage
[8] Thomas, C. E. Fuel cell and battery electric vehicles compared. International Journal of Hydrogen Energy 34, 6005–6020 (2009)
[9] IEA, Clean Energy Ministerial, and Electric Vehicles Initiative (EVI) (2021). “Global EV Outlook 2021: Accelerating ambitions despite the pandemic”.
Discover more from evoiding
Subscribe to get the latest posts sent to your email.
